Inhaltsverzeichnis
Investigations on a possible formation and avoidance of chromate in leather 1999
Dr. Christiane Hauber and Dr. Heinz-Peter Germann, Lederinstitut Gerberschule Reutlingen, Germany
Chrome tannage is the most important tanning method representing over 80% of leather production. It is very versatile and no change is expected in the foreseeable future.
Chromate[Cr(VI)] has sometimes been detected in chrome tanned leathers. This is unexpected because in the presence of a high proportion of organic matter and low pH, chrome(VI) is unstable, and chrome(III) is produced by reduction.
Various possibilities for the presence of Cr(VI) have been cited:
Neutralisation ofthe wet-blue, as during this treatment the acid present is neutralised and the pH raised.
Any dyeing pre-treatment in order to level the dyeing and improve penetration. Ammonia is sometimes used, often in an overnight float to promote through dyeing.
Drying and intermediate drying might provide conditions for the formation of Cr(VI). It is also thought possible that heat and light can oxidise Cr(III) to Cr(VI).
The influence of ratty acids in fatliquors cannot be ignored.
Details of the investigation
In a series of investigations clothing, upholstery and water resistant shoe upper leathers were produced to cover the main leather types. The pelt was chrome tanned to the wet-blue stage, neutralised in various ways and taken to the crust. After additions to give good dye penetration, the leathers were dyed. Redox potentials were determined) at the individual process stages in order to assess the oxidative or reductive properties of the floats. In addition, 5 and 10 mg/1 chromate was added and the influence of the reduction measured according to DIN 38405.
The effects of retannage and fetliquoring were also investigated. The Cr(VI) content of the undyed/dyed leathers in their intermediate states were analysed photometrically according to DIN 53314 using the reaction between Cr(VI) and 1.5 diphenyl carbazide.
The leather was analysed in:
- the dried aired-off State.
- after heating at 80°C for 24 hours in a drying chamber.
- after subjection to UV radiation for 48 hours in a Xenotest apparatus.
It should be noted that the conditions created by treatments (b) and © are extreme and unlikely to be met in normal use.
The influence of neutralising
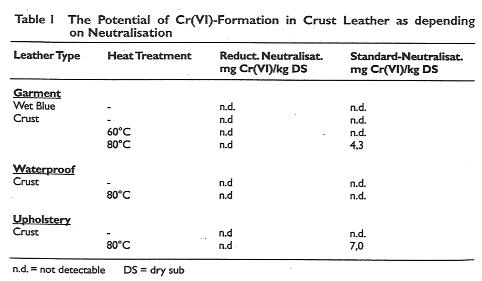
The redox and chromate reduction potential in the neutralisation of wet-blue for upholstery crust, clothing and water resistant shoe-upper were assessed.
The wet-blue was neutralised conventionally, using sodium formate and sodium bicarbonate and compared with a reducing auxiliary. The reducing potential was also checked by the addition of 5 and 10 mg/1 Cr(VI) to the float.
No relationship could be established between the chromate reduction property ofthe float and the Cr(VI) content ofthe leather produced. The Cr(VI) content of the various crust leathers were below 3mg/kg, the detection limit based on dry substance.
However, when the results involving heating at 80°C for 24 hours are considered, it can be seen that in isolated cases ofthe Standard neutralisation Cr(VI) is detectable. This is shown in Table 1.
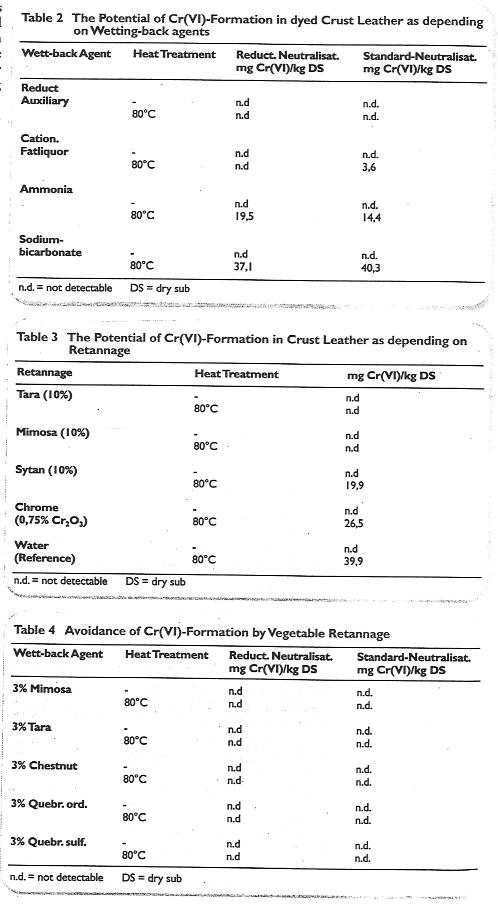
The influence of wetting back agents before dyeing crust leathers
The reduction tendency of the bath before dyeing was investigated on different leathers with various wetting back agents. The results are given of dyeings on crust upholstery leathers with:
- standard neutralisation using sodium formate and sodium bicarbonate
- the addition of reducing auxiliary agents to the Standard neutralisation.
It was found that the Cr(VI) content of all the air dried leathers was below the detection limit. However, after heating at 80°C for 24 hours, the ammonia or sodium bicarbonate treated samples contained chromate as shown in Table 2.
Table 1:
In further investigations a reducing auxiliary was used as a wetting back agent before dyeing, and in another experiment ammonia. The acidification for fixation was carried out at die end of die dyeing process with ascorbic acid instead of fbrmic acid normally used. This was to ensure an especially high reducing ability of die acidification float. Despite the higher reducing properties of both the wetting back float and acidification, Cr(VI) formation could not be prevented in subsequent heating at 80°C for 24 hours.
The influence of vegetable retannage
Experiments with water resistant shoe upper leather showed that the crust and dyed leather even after extreme treatment showed no Cr(VI) content. The main difference from the upholstery and clothing recipes was the use of mimosa. Experiments were therefore carried out with different retannages. Wet blue leathers were neutralised conventionally with sodium formate/bicarbonate and retanned with either 10% tara, 10% mimosa, 10% synthetic tanning agent or a chrome containing tanning material. A blank test was made without retannage. All were given 8% fatliquor containing natural and synthetic components and finished conventionally into crust. The findings are presented in Table 3.
As a result of diese findings further retannages were applied at 3% offers of mimosa, tara, chestnut, quebracho (ordinary and sulflted). Standard neutralisation and neutralisation with a reducing auxiliary were used and the findings are shown in Table 4.
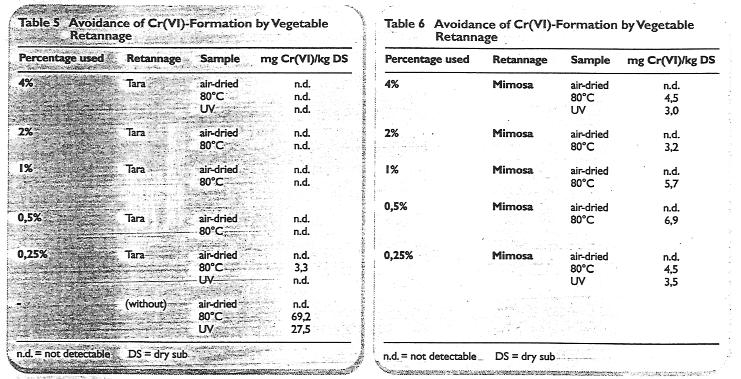
For many leather types it is not possible to add large amounts of vegetable retan therefore lower offers were made to determine the level at which a reduction in chromate production was noticeable.
The results of offers of tara and mimosa when neutralised and fatliquored with 8.0% fatliquor containing natural and synthetic components are given in Table 5 and Table 6.
It is dear that after extreme conditions -heat and UV exposure - the mimosa did not prevent chromate formation. Tara, however, was better and all results were successfiil in achieving the avoidance of Cr(VI) formation.
The „blank“ showed that although under normal drying conditions Cr(Vl) was not deteaed, under extreme exposure conditions rektively high Cr(VI) values resulted.
Table 2 to 4:
Table 5 to 6:
The influence of fatliquors
From the results presented, and other investigations, there was evidence of a linkage between Cr(VI) formation, neutaralisation and wetting back agents. In addition, with the same retannage differences could occur with various fatliquors in the amounts of chromate found. It was thought that the influence of the fatliquoring could be due to the effetcs of unsatuarated fats on Cr(III) oxidation. It was considered that raw fat materialls of highm iodine value (more unsaturation) could play a role in Cr(VI) formation.
Experiments were therefore undertanken, after a chrome retannage and normal neutralisation with sodium formate/bicarbonate to a maximum pH of 5,5. Fatliquors were offered at 10% includig fish oils of different iodine value. These findings are oppresented as Table 7.
No differences were found between sulfited fish oils with different iodine values when air dried or drid at 80C.
However, with lower iodine values, the chromate contents of leathers after UV exposure were distinctly lower.
Table 7 and Table 8:
The effect of oleiic acid and other variables
An overwview pf the fatliquor types used in this investigation is presented in Table 8.
This shows that as well as the degree of unsaturation, other fatty compunds - in particular oleic acid - affect thje formation of Cr(VI). Oleeic acid contains only a single duble bond but can affect the oxidation of Cr(III) to Cr(VI). The analytical values show that no air ried leathers had detectaeble chromate. After leating the synthetic fatliquor(without unsaturated components) was advantageos but also that in the presence of eoleicm acid chromate formation was a possibility.
It also shows thaht sulfonated and sulfited fishoils increase the tendeny to Cr(VI) formation under the extreme conditions of heat and UV exposure.
The use of some lecithin based fatliquors also appears undesirable, being uses in the fatliquoring of clothing leathers.
The effects of retanning agents and fatliquors
The effects of polymer and mimosa retannages with fatliquors
It has been shown when using certain retanning materials and fatliquorsthere is no chromate formation even after extreme conditioning.
In a fiirther series of experiments, the combination of specific fatliquorsand retannages was examined. The results of combinations of fatliquorswith a polymer or mimosa retannage is given in Table 9.
It can be seen that:
- in the Variation without fatliquör, the mimosa retannage was inferior to the polymer retannage.
- in the combination with fatliquorse.g. sulfited fish oil, the combinations with mimosa generally led to lower Cr(Vl) values.
The effects of tara with different fatliquor combinations
Of the vegetable retannages, tara has particularly good effect in avoiding the formation of chromate. It was therefore used in combination with the fatliquor types used to 2ssess the effects of oleic acid and other variables, (finding given as Table 8) to determine the amount of tara required to suppress chromate formation.
The wet blue were therefore neutralised in a similar manner, then retanned with varying offers of tara, washed and then fatliquored using a 10% offer. The findings are presented in Table 10.
It can be seen that:
- The only air dried leather showing a trace of Cr(VI) was produced using an oleic acid containing syntheric fatliquor without tara.
- That low levels of tara was unable to prevent the formation of chromate when using sulfited or sulfonated fish oils alone under extreme conditions of heat and UV exposure.
- In the case of tara at 2.0% offer and sulfited fish oil, traces of chromate were only detected after UV exposure.
- In all other cases, the tara retannage provided avoidance of all chromate formation.
Table 9 and 10:
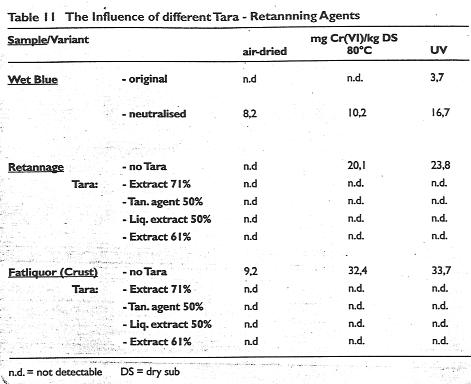
The influence of different types of tara
Different types of tara were investigated in the production of automobile upholstery leather being 71% and 50% tanning extract, 50% tan content liquid extract and a further extract with 61% tan content. In these investigations a limited amount of conventipnal fatliquor was used during neutralisation and similarly in the retannage. The findings are presented in Table 11.
It can be seen that:
- the wet blue showed a chromate after exposure to UV.
- the leather after neutralisation, retannage without tara and fatliquoring had a noticeable chromate content after heat and UV exposure.
- all of the 4 tara extract treated samples showed no detectable chromate.
The influence of storage
Prevailing wisdom suggests that storage could increase chromate content. Some leathers - dothing, furniture, and shoe upper leathers - were stored for two months and reanalysed for chromate.
It was found that whilst some samples decreased, others increased compared with the original results.
However, where vegetable retanning agents and fatliquors were used that showed no formation of Cr(VI) in the fresh State, no presence was detected after storage.
These observadons could account for the variability of analytical results of different workers that have been interpreted as inadequacies of analytical methods up to now. The results indicate an ongoing interchange between chrome complexes and other components in the leather with the tannage.
The influence of mechanical processes
In buffing, for example, in the producdon of nubuck, there is at least a surface heating effect. This was investigated to see if there was an influence on chromate formation.
No chromate formation was detected in leathers which were chromate free before buffing. Where there was detectable chromate present, a slight increase was found, depending on the applied energy (rotation speed).
In vacuum drying there is a short heat application but no chromate formation occurs in leathers which are chromate free.
Table 11:
Summary of findings
- Measurement of redox potential of process liquors show no linkage to chromate formation.
- Measurement of chrome reduction properties of the individual process liquors showed no connection with the chromate content of the processed leather.
- The partial replacement of normal neutralisation materials by a reducing auxiliary, was found to reduce chromate formation.
- The use of a reducing auxiliary prior to dyeing is recommended. Stronger alkaline agents such as ammonia or sodium bicarbonate should be avoided.
- The type of fatliquoring agent has a profound influence on the possibilities of chromate formation.
The greatest effect is shown by:
- the dassical sulfonated and sulfited fish oils
- products with simple or multiple unsaturated free or esterified fatty acids.
Natural or synthetic fatliqoring agents wnich do not contain diese substances, do not lead to chromate formation.
- Synthetic and polymeric retanning materials are not conducive to the formation of Cr(VI). In critical leathers diese products do not prevent the formation of Cr(VI) on extreme heat and UV exposure. Vegetable retannage plays a significant part in avoiding chromate formation.In the research mimosa, quebracho, chestnut and tara showed a positive influence in leathers exposed to extremes of heat and UV exposure. Tara is particularly effective, where on some leathers even an addition of 0.25% produced suppression of chromate formation.
- Both reductions and increases in chromate content of the leather were found after 2 months storage. Where vegetable retanning agents and fatliquors were used that showed no formation of Cr(VI) in the fresh State, no presence was detected after storage.
- When leathers contain Cr(VI), buffing can give rise to an increase in Cr(VI). This does not occur in leadiers which show no Cr(VI) before buffing.
- Vacuum drying has no influence on Cr(VI) formation.
References
- Cory, N.J.: JALCA 92(1997), 119.
- Gruppo Soci veneti AICC: Konferenzbericht (1998)
- Nickolaus G.: XI International Congress on the Leather, Shoe Industries proceedings, Budapest (1998)
- PüntenerA.: JALCA 91 (1998), 126
- Shi B.: XXIV IULTCS Congress proceedings, London 1997
- World Leather 11 (1998) Nov, 5.
- DIN 53314: Bestimmung des Chrom(Vl)-Gehaltes in Ledern.
- DIN 38405: Photometrische Bestimmung von Chrom(Vl) mittels 1.5-Diphenyl-carbazid (D24)
- DIN 38404: Physikalische und physikalisch-chemische Kenngrössen (Gruppe Q-Bestimrnung der Redox-Spannung.
Selective extracts ofthis is notpermitted without written permission ofther publisher.
Publication:
C. Hauber, H.-P. Germann, Investigations on a possible formation and avoidance of chromate in leather 1999, World Leather 12, 10/1999, No. 6, p. 73 - 80
Kategorien:
Quellenangabe:
Zitierpflicht und Verwendung / kommerzielle Nutzung
Bei der Verwendung von Inhalten aus Lederpedia.de besteht eine Zitierpflicht gemäß Lizenz CC Attribution-Share Alike 4.0 International. Informationen dazu finden Sie hier Zitierpflicht bei Verwendung von Inhalten aus Lederpedia.de. Für die kommerzielle Nutzung von Inhalten aus Lederpedia.de muss zuvor eine schriftliche Zustimmung (Anfrage via Kontaktformular) zwingend erfolgen.
www.Lederpedia.de - Lederpedia - Lederwiki - Lederlexikon
Eine freie Enzyklopädie und Informationsseite über Leder, Ledertechnik, Lederbegriffe, Lederpflege, Lederreinigung, Lederverarbeitung, Lederherstellung und Ledertechnologie